Horseradish Peroxidase (HRP, EC 1.11.1.7) is a heme-containing glycoprotein derived from the roots of horseradish (Armoracia rusticana). As a classical biocatalytic tool enzyme, HRP is widely utilized in medical diagnostics, molecular biology, and environmental remediation due to its high catalytic activity, robust stability, and excellent labeling compatibility. This article provides a systematic overview of HRP's core characteristics, manufacturing processes, and application scenarios for life science professionals.

Part I: Core Characteristics of HRP
1.1 Structure and Catalytic Mechanism
HRP is a heme-containing glycoprotein with a molecular weight of approximately 40 kDa, comprising 308 amino acid residues and 8 N-glycosylation sites (carbohydrate content: 18-22%). The prosthetic group consists of heme and two Ca²⁺ ions, with heme serving as the catalytic core. Its primary function is to catalyze hydrogen peroxide-mediated oxidation of hydrogen donors, following the reaction: Donor + H₂O₂ → Oxidized donor + 2H₂O. The enzyme remains unconsumed during catalysis, exhibiting high efficiency and strong substrate specificity.
1.2 Key Characteristics and Purity Indicators
Core characteristics of HRP include: stability at room temperature for several weeks; retention of activity at 63°C for 15 minutes; superior specific enzyme activity; and specific recognition of H₂O₂ and particular hydrogen donors.
HRP exhibits maximum absorbance at 403 nm (Soret band) and 275 nm (protein band). The ratio of absorbance at 403 nm to 275 nm (A₄₀₃/A₂₇₅) serves as the critical purity indicator. This methodology was developed by German scientist Reinheit Zahl, and the ratio is commonly referred to as the RZ value (Reinheit Zahl). HRP purity grades are classified as follows:
- RZ > 3.0: High-purity grade for immunological assays
- RZ > 2.0: Clinical chemistry grade
- RZ > 1.0: General diagnostic test strips
- RZ > 0.6: Low-precision applications
HRP contains at least seven isoenzymes, with isoenzyme C1A (UniProt: P00433) being the most abundant, exhibiting optimal activity, and most widely applied.
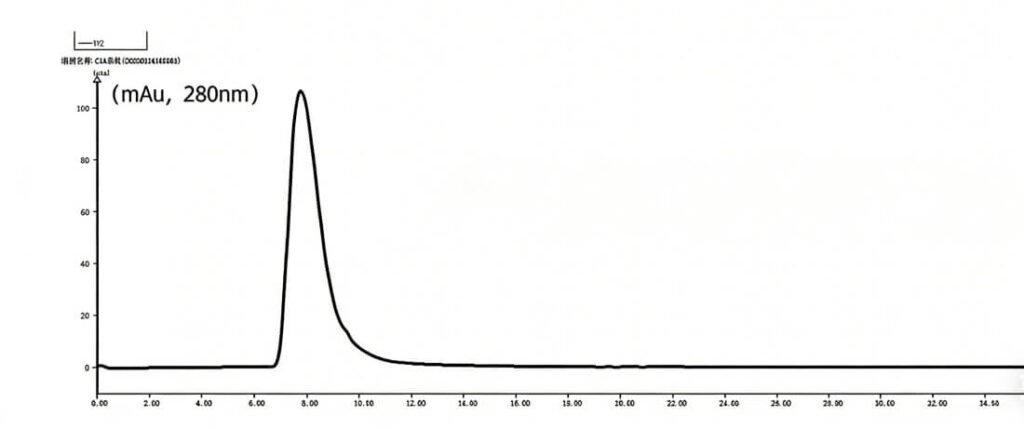
Tinzyme HRP demonstrates 100% isoenzyme C1A content as verified by liquid chromatography analysis.
1.3 Product Forms and Storage Requirements
Commercial HRP is available in two primary formats: lyophilized powder and liquid enzyme preparation. Lyophilized powder offers extended shelf life and convenient transportation, requiring reconstitution in appropriate buffer prior to use. Liquid formulations are ready-to-use and require storage at 2-8°C. All HRP products must be protected from elevated temperatures and inhibitors such as cyanides and fluorides to prevent activity loss.

Part II: HRP Manufacturing Technologies
The primary objective of HRP production is to obtain high-purity, high-activity enzyme preparations. Current mainstream production methodologies are divided into natural extraction and heterologous recombinant expression, which complement each other to meet diverse manufacturing requirements.
2.1 Natural Extraction Method
The extraction process utilizes fresh horseradish roots as raw material, proceeding through washing and homogenization, low-temperature buffer extraction, ammonium sulfate/acetone fractional precipitation, dialysis desalting, zinc ion purification, and lyophilization to obtain finished HRP product. This method offers readily available raw materials, straightforward processing, and lower costs, suitable for small-scale production. However, it presents limitations including restricted yield, inconsistent purity, and susceptibility to agricultural cultivation conditions.
2.2 Recombinant Expression and Advanced Technologies
Heterologous recombinant expression represents the mainstream approach for large-scale production. The core workflow comprises: HRP gene cloning and optimization → host cell transformation (E. coli, Pichia pastoris, etc.) → fermentation culture → chromatographic purification → activity assessment. This methodology delivers high yield, consistent purity, and enables enzyme activity enhancement through genetic optimization. The primary limitations involve complex processing and higher initial capital investment.
Recombinant plant expression represents an advanced HRP production technology. This approach involves cloning the HRP gene into appropriate vectors and introducing the construct into plant tissues (e.g., leaves), enabling expression during plant growth. The core workflow: HRP gene cloning and optimization → gene vector construction → plant tissue transformation (e.g., leaves) → hydroponic cultivation → leaf harvest → tissue homogenization → chromatographic purification → activity assessment.
This method offers significant advantages including high yield, superior activity, exceptional purity, and scalability for expanded production.
Cell-free protein synthesis (CFPS) systems are increasingly adopted, achieving synchronous synthesis of HRP and prosthetic groups through in vitro modular assembly, further enhancing activity and yield while reducing production costs.
2.3 Quality Control Standards
Critical quality parameters for finished products include RZ value (purity), specific enzyme activity (catalytic efficiency), molecular weight, and contaminant protein content. Primary analytical methods encompass spectrophotometry (for RZ value and enzyme activity determination) and electrophoresis (for purity and molecular weight verification). Only products meeting established specifications are approved for commercial distribution.
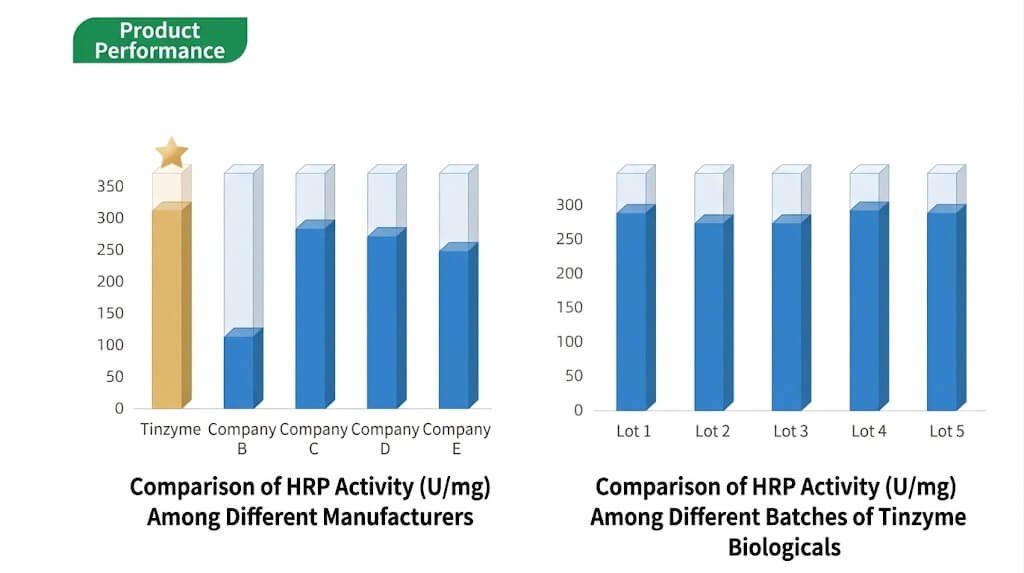
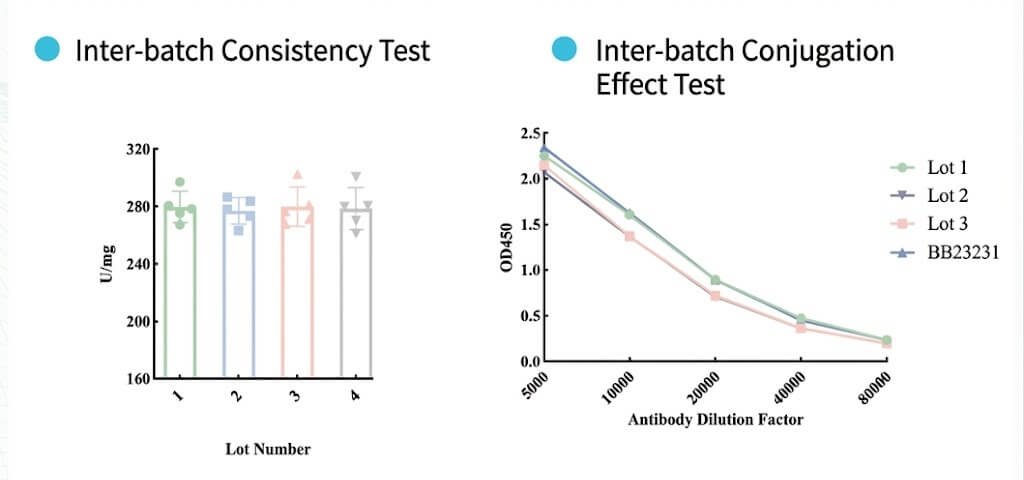
Comparative testing demonstrates that Tinzyme HRP delivers superior performance with minimal batch-to-batch variation.
Part III: Comprehensive Application Areas
3.1 Medical and Clinical Diagnostics
HRP finds its most extensive applications in medical diagnostics, with core applications including:
Enzyme-Linked Immunosorbent Assay (ELISA): HRP-conjugated antibodies combined with TMB, OPD, or other chromogenic substrates enable quantitative detection of antigens/antibodies for infectious disease and tumor marker screening.
Immunohistochemistry (IHC) and Immunofluorescence (IF): HRP-labeled antibodies facilitate localization of target proteins in tissue sections, supporting pathological diagnosis.
Western Blot: HRP-conjugated secondary antibodies enable detection of target protein expression levels through chemiluminescence (luminol substrates) or chromogenic development.
Additionally, HRP serves as a superior alternative to colloidal gold in home diagnostic test strips, significantly enhancing detection sensitivity.
3.2 Research Applications
In molecular biology, HRP functions as a reporter enzyme for gene detection, nucleic acid hybridization, and gene cloning/expression analysis. In biochemistry, HRP serves as a model enzyme for investigating oxidation-reduction reaction mechanisms, providing foundational insights for novel enzyme development.
3.3 Environmental and Industrial Applications
Environmental remediation applications leverage HRP to catalyze degradation of phenolic and aromatic contaminants in industrial wastewater and soil. In food industry applications, HRP enables hydrogen peroxide residue detection and polyphenol analysis for food safety assurance. HRP is additionally employed in polymer synthesis and biosensor fabrication.
3.4 Application Guidelines
Appropriate substrate selection is essential for specific applications: TMB and DAB for routine chromogenic detection; luminol for high-sensitivity detection; Amplex Red for fluorescent detection. Contact with inhibitors including cyanides, fluorides, and azides must be avoided to prevent enzyme inactivation.
Part IV: Future Perspectives
As a classical biocatalytic tool enzyme, HRP maintains its prominent position after decades of widespread application. Future developments will focus on optimization of heterologous recombinant technologies and cell-free synthesis systems to reduce production costs and enable large-scale, high-quality manufacturing. Genetic engineering approaches will further optimize HRP structure and performance, expanding applications in precision medicine and next-generation biosensors, thereby sustaining its core value in biotechnology.
Tinzyme HRP Advantages:
• Manufactured through recombinant expression technology with 100% isoenzyme C1A content, delivering specific catalytic activity ideal for high-sensitivity diagnostic reagents (e.g., ELISA) and biosensor applications
• Produced via plant expression systems, completely free from exogenous microorganisms, viral contaminants, and animal-derived components, eliminating pathogenic risk and ensuring full compliance with FDA/EMA stringent regulatory requirements for in vitro diagnostic reagents
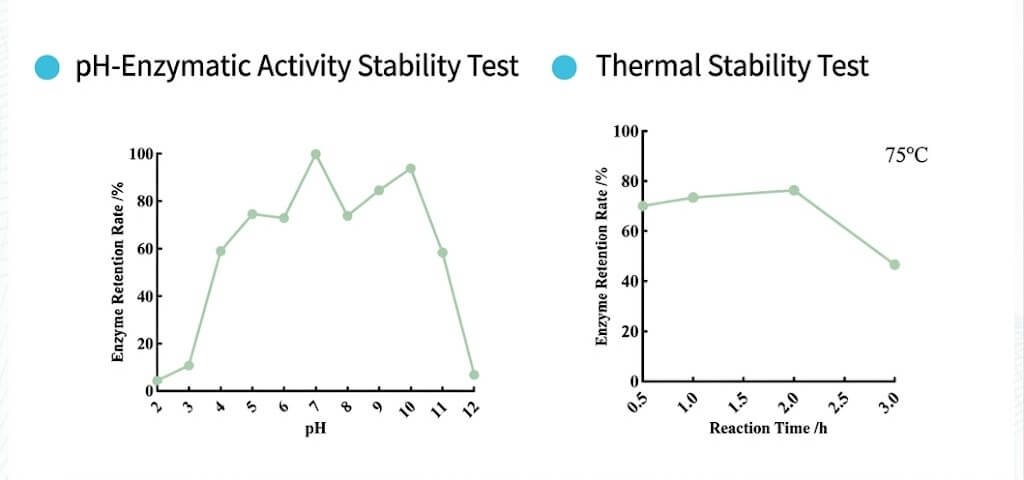
• Engineered through directed evolution to maintain activity across broad pH range (3-10) and elevated temperatures (≤60°C), accommodating demanding reaction conditions
